Emission spectrum examples11/8/2023  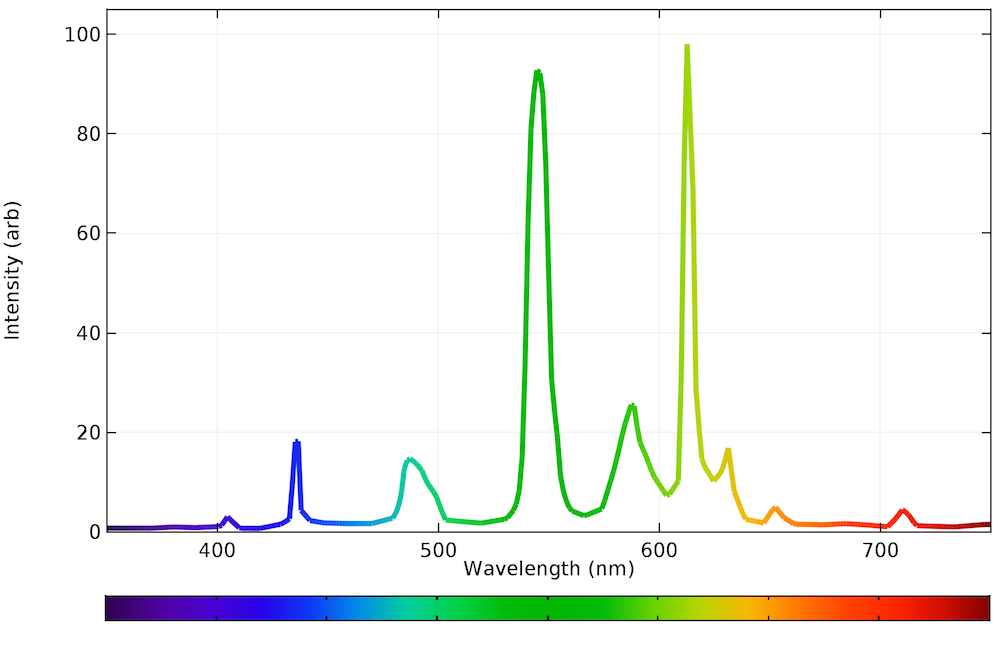
An electron in a starting position that is farther from the nucleus already has more energy because it must compensate for the great distance that separates it from the nucleus. Energy must be added to this electron for it to “jump” to a higher orbital, and the further away it jumps, the more energy it would need. The model further specified that electrons orbit the nucleus at well defined distances from the nucleus.Īn electron orbiting near the nucleus has relatively little energy because it is held in its orbital by the strong attractive force exerted by the nucleus. According to Bohr, the atom consists of negatively charged electrons that orbit around a positively charged nucleus. In 1913, the Danish physicist Niels Henrik David Bohr integrated the advances of Planck and Einstein into his new model of the atom to explain how matter could emit light in the form of particles. He concluded that waves with long wavelengths, like red light, transport small amounts of energy, whereas those with short wavelengths, like violet light, transport more energy. 
Einstein not only supported Planck’s idea, he went one step further: he proposed that light behaved as a particle (a photon) but also retained its wave character and must therefore be considered as both a particle and a wave.įurthermore, Einstein established that the energy of a photon is related to the wavelength of the radiation. The debate finally came to a head – and to a resolution – when the German physicist Albert Einstein entered the scene in 1905. This proposal ran contrary to all contemporary knowledge about light at that time. In his theory, Planck maintained that light could only be emitted as small packets of energy that he named “quanta” (later known as “photons”). Planck noted that it was impossible to solve the problem using existing principles of physics and proceeded to develop a revolutionary theory that marked nothing less than the beginning of modern physics. It was only in 1900 that the German physicist Max Planck would provide part of the answer. Many researchers attempted to explain how matter could generate an emission spectrum but without success. In 1859, the German physicist Gustav Robert Kirchhoff called this type of spectrum an “emission spectrum”. Despite this lack of understanding, they nonetheless knew how to separate the light emitted by a gas into a spectrum that was diagnostic of the chemical elements contained in the gas. I think this is when white light is used that you get an Absorption Spectra.By the end of the 1800’s, scientists were observing this phenomenon in their laboratories but could not explain it. All the colors of the Absorption Spectra do make it kind of confusing. And these are being absorbed (with emphasis on blue). Actually, if you just burned hydrogen and looked at its spectra, you would get the Emission Spectra and not the Absorption Spectra, and this Emission Spectra would only show the bunch of blue lines, one purple line, and one red line. All the other colors shown are just part of the natural light being shown down on the element. This is the color that will be the opposite of the flame color on the color wheel. 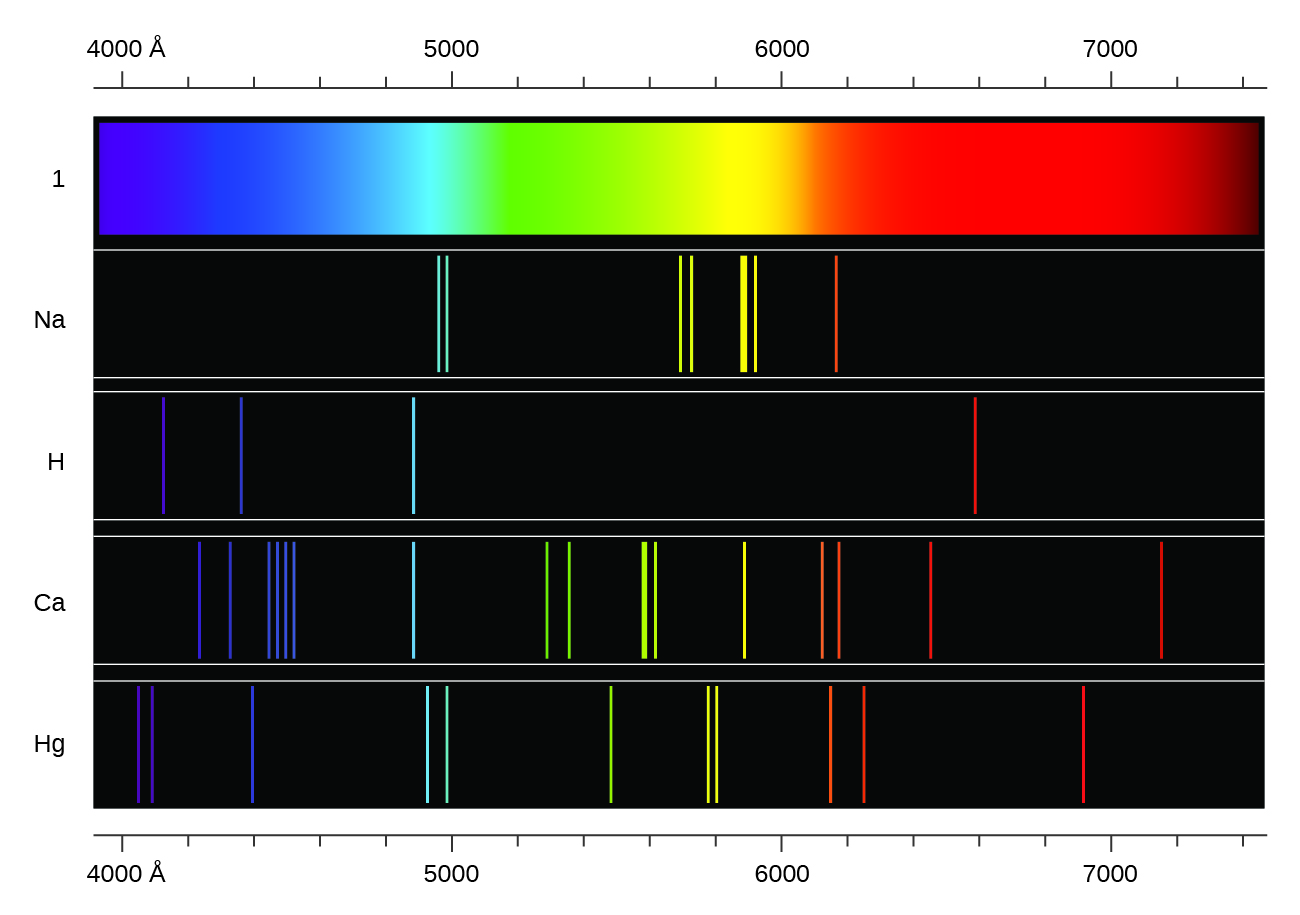
Remember, always look at the color area on the rainbow that is blacked out the most. So if blue is being absorbed, the opposite color would be transmitted and this color is orange. However, there are MORE dark lines in the blue region. If you look at the lines for hydrogen blue, purple, and red are being absorbed. Therefore, all the other colors would be absorbed. (This would be orange.) The element hydrogen turns orange when being burned and this color is transmitted to us. This means that if there is a big dark band where blue would be, then the opposite color to blue on the color wheel is being transmitted. You are supposed to look at the dark areas of the absorption spectra and those dark areas indicate that the color which would be there is being absorbed. I think both the absorption and emission lines are showing which colors are being absorbed. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
